Exodus Paste
This treatment applies to the following species:(pyrantel pamoate)
ANADA 200-350, Approved By FDA
EQUINE ANTHELMINTIC
FOR ORAL USE IN HORSES ONLY
Apple Flavored!
Exodus® Paste (pyrantel pamoate) is a pale yellow to buff paste containing 43.9% w/w pyrantel pamoate in an inert vehicle. Each 23.6 g single-dose syringe contains 3.6 grams pyrantel base in 23.6 grams paste. Each 47.2 g multi-dose syringe contains 7.2 grams pyrantel base in 47.2 grams paste. Each milliliter contains 171 milligrams pyrantel base as pyrantel pamoate.
READ ENTIRE CARTON CAREFULLY BEFORE USING THIS PRODUCT
Composition
Pyrantel pamoate is a compound belonging to a family classified chemically as tetrahydropyrimidines. It is a yellow, water-insoluble crystalline salt of the tetrahydropyrimidine base and pamoic acid containing 34.7% base activity. The chemical structure and name are given below.
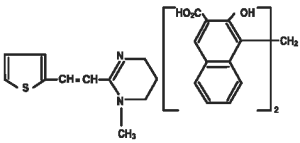
Chemical Name:
(E)-1,4,5,6-tetrahydro-1-methyl-2-[2-(2-thienyl)-vinyl]-pyrimidine 4,4’ methylenebis [3-hydroxy-2-naphtholate] (1:1)
Exodus Paste Indications
For the removal and control of mature infections of large strongyles (Strongylus vulgaris, S. edentatus, S. equinus); small strongyles; pinworms (Oxyuris equi); and large roundworms (Parascaris equorum) in horses and ponies. Consult your veterinarian for assistance in the diagnosis, treatment, and control of parasitism.
Dosage And Treatment
Exodus® Paste (pyrantel pamoate) is to be administered as a single oral dose of 3 milligrams pyrantel base per pound of body weight. The 23.6 g syringe has four weight mark increments. The 47.2 g syringe has eight weight mark increments. Each weight mark indicates the recommended dose for 300 pounds of body weight. One-half 47.2 g syringe treats a 1200 lb horse.
DOSAGE for Single-Dose 23.6 g Syringe:
|
Body Weight Range |
Quantity |
mg Pyrantel Base |
|
up to 300 lb |
1/4 syringe |
900 mg |
|
301 to 600 lb |
1/2 syringe |
1800 mg |
|
601 to 900 lb |
3/4 syringe |
2700 mg |
|
901 to 1200 lb |
1 full syringe |
3600 mg |
DOSAGE for Multi-Dose 47.2 g Syringe:
|
Body Weight Range |
Quantity |
mg Pyrantel Base |
|
up to 300 lb |
1/8 syringe |
900 mg |
|
301 to 600 lb |
1/4 syringe |
1800 mg |
|
601 to 900 lb |
3/8 syringe |
2700 mg |
|
901 to 1200 lb |
1/2 syringe |
3600 mg |
NOTE: Position ring-gauge over appropriate mark on plunger.
For maximum control of parasitism, it is recommended that foals (2-8 months of age) be dosed every 4 weeks. To minimize the potential source of infection that the mare may pose to the foal, the mare should be treated 1 month prior to anticipated foaling date followed by re-treatment 10 days to 2 weeks after birth of foal. Horses and ponies over 8 months of age should be routinely dosed every 6 weeks.
It is recommended that severely debilitated animals not be treated with this preparation.
Administration
After removing the cap, the paste should be deposited on the dorsum of the tongue. Introduce the nozzle end of the syringe at the corner of the mouth. Direct the syringe backwards and depress the plunger to deposit the paste onto the tongue. Given in this manner, it is unlikely that rejection of the paste will occur.
Raising the horse’s head sometimes assists in the swallowing process. When only part of the paste has been used, replace the cap on the syringe nozzle.
Efficacy
Critical (worm count) studies in horses demonstrated that pyrantel pamoate administered at the recommended dosage was efficacious against mature infections of Strongylus vulgaris (>90%), S. edentatus (69%), S. equinus (>90%), Oxyuris equi (81%), Parascaris equorum (>90%), and small strongyles (>90%).
WARNING: Do not use in horses intended for human consumption.
KEEP OUT OF REACH OF CHILDREN
FOR ORAL USE ONLY
FOR ANIMAL USE ONLY
STORE AT 20°C TO 25°C (68°F TO 77°F); excursions permitted between 15°C and 30°C (between 59°F to 86°F).
Manufactured by: Bimeda-MTC Animal Health Inc., Cambridge, ON Canada N3C 2W4
Manufactured for: Bimeda, Inc., Le Sueur, MN 56058
www.bimedaus.com
N.A. Corp. Address: Bimeda, Inc., One Tower Lane, Oakbrook Terrace, IL 60181
® Exodus is a registered trademark of Bimeda, Inc.
|
Net Wt. |
Product No. |
|
|
23.6 g (0.83 oz) Single-Dose |
1EXO001 |
8PYR003H REV. 10/14 |
|
47.2 g Multi-Dose |
1EXO002 |
8PYR006C Rev. 12/13 8PYR005F Rev. 04/14 |
CPN: 1399063.10
Div. Cross Vetpharm Group, Ltd.
475 NORTH MARTINGALE ROAD, SUITE 1200, SCHAUMBURG, IL, 60173
| Telephone: | 630-928-0361 | |
| Fax: | 630-928-0362 | |
| Website: | www.bimedaus.com | |
| Email: | us-sales@bimeda.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-03-02
