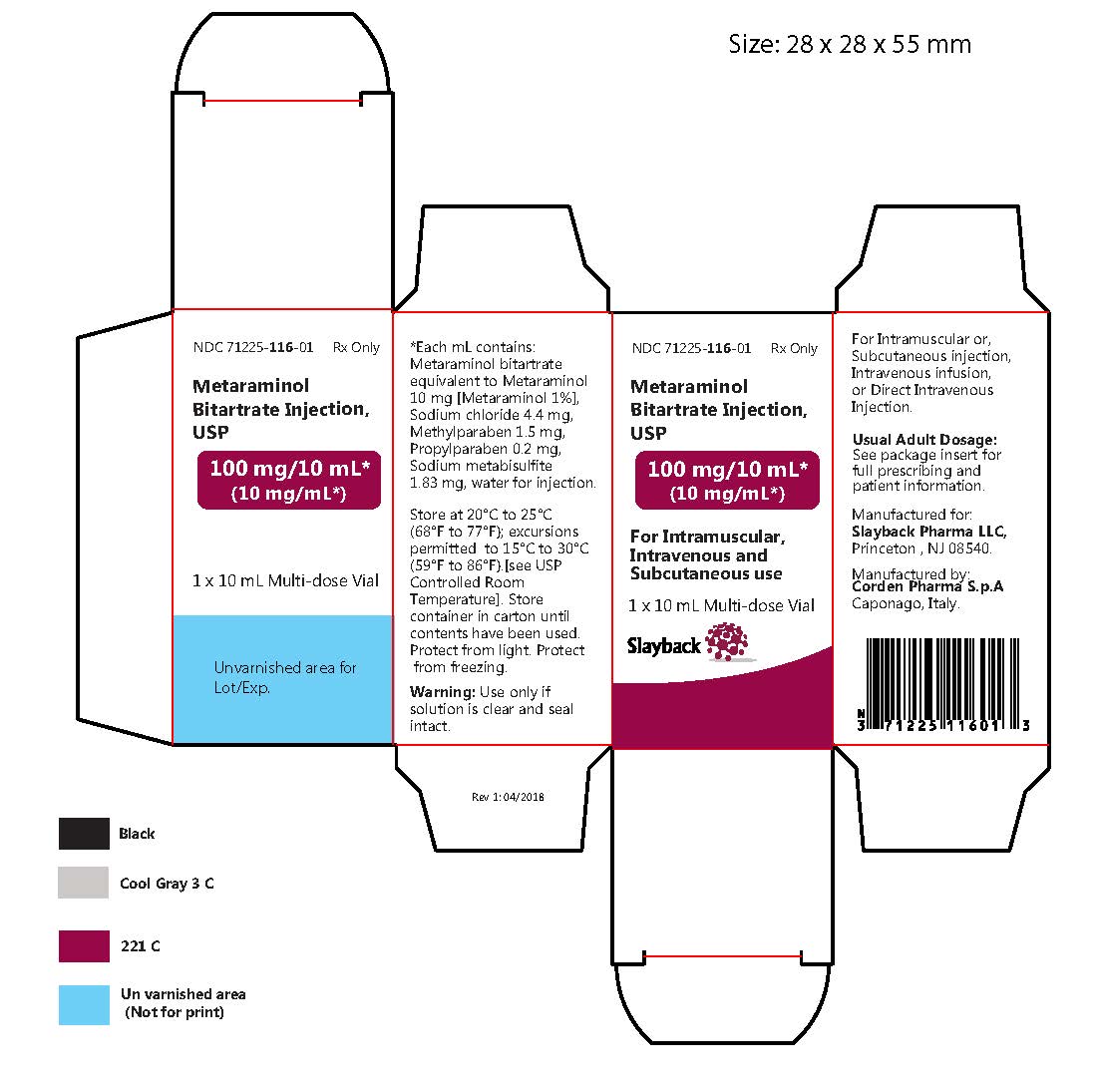
Metaraminol Bitartrate Injection: Package Insert / Prescribing Info
Package insert / product label
Dosage form: injection
Drug class: Vasopressors
Medically reviewed by Drugs.com. Last updated on Jul 11, 2024.
On This Page
Metaraminol Bitartrate Injection Description
Metaraminol bitartrate is a potent sympathomimetic amine that increases both systolic and diastolic blood pressure.
Metaraminol bitartrate is [R-(R*,S*)]-α-(1-aminoethyl)-3-hydroxybenzenemethanol [R-(R*,R*)]-2,3-dihydroxybutanedioate(1:1) (salt), which is levorotatory. Its empirical formula is C
9H
13NO
2•C
4H
6O
6and its structural formula is:
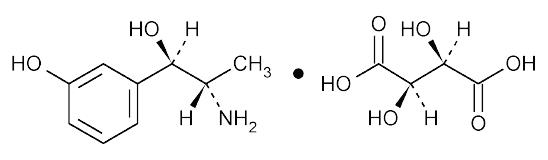
Metaraminol bitartrate is a white, crystalline powder with a molecular weight of 317.29, is freely soluble in water, slightly soluble in alcohol, and practically insoluble in chloroform and in ether.
Metaraminol bitartrate injection, USP is a sterile solution. Each mL contains:
Metaraminol bitartrate
equivalent to
metaraminol ..... 10 mg
Inactive ingredients:
Sodium chloride ..... 4.4 mg
Water for Injection q.s. ad ..... 1 mL
Methylparaben 0.15%, propylparaben 0.02%, and sodium metabisulfite 0.183% added as preservatives.
Metaraminol Bitartrate Injection - Clinical Pharmacology
The pressor effect of metaraminol bitartrate injection begins in 1 to 2 minutes after intravenous infusion, in about 10 minutes after intramuscular injection, and in 5 to 20 minutes after subcutaneous injection. The effect lasts from about 20 minutes to one hour. Metaraminol bitartrate injection has a positive inotropic effect on the heart and a peripheral vasoconstrictor action.
Renal, coronary, and cerebral blood flow are a function of perfusion pressure and regional resistance. In patients with insufficient or failing vasoconstriction, there is additional advantage to the peripheral action of metaraminol bitartrate injection, but in most patients with shock, vasoconstriction is adequate and any further increase is unnecessary. Blood flow to vital organs may decrease with metaraminol bitartrate injection if regional resistance increases excessively.
The pressor effect of metaraminol bitartrate injection is decreased but not reversed by alpha-adrenergic blocking agents. Primary or secondary fall in blood pressure and tachyphylactic response to repeated use are uncommon.
Indications and Usage for Metaraminol Bitartrate Injection
Metaraminol bitartrate injection is indicated for prevention and treatment of the acute hypotensive state occurring with spinal anesthesia. It is also indicated as adjunctive treatment of hypotension due to hemorrhage, reactions to medications, surgical complications, and shock associated with brain damage due to trauma or tumor.
Contraindications
Use of metaraminol bitartrate injection with cyclopropane or halothane anesthesia should be avoided, unless clinical circumstances demand such use.
Hypersensitivity to any component of this product, including sulfites (see WARNINGS).
Warnings
Use of sympathomimetic amines with monoamine oxidase inhibitors or tricyclic antidepressants may result in potentiation of the pressor effect. (See PRECAUTIONS, Drug Interactions.)
Metaraminol bitartrate injection contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
Precautions
GENERAL PRECAUTIONS
Caution should be used to avoid excessive blood pressure response. Rapidly induced hypertensive responses have been reported to cause acute pulmonary edema, arrhythmias, cerebral hemorrhage, or cardiac arrest.
Patients with cirrhosis should be treated with caution, with adequate restoration of electrolytes if diuresis ensues. Fatal ventricular arrhythmia was reported in one patient with Laennec's cirrhosis while receiving metaraminol bitartrate. In several instances, ventricular extrasystoles that appeared during infusion of this vasopressor subsided promptly when the rate of infusion was reduced.
With the prolonged action of metaraminol bitartrate injection, a cumulative effect is possible. If there is an excessive vasopressor response there may be a prolonged elevation of blood pressure even after discontinuation of therapy.
When vasopressor amines are used for long periods, the resulting vasoconstriction may prevent adequate expansion of circulating volume and may cause perpetuation of shock. There is evidence that plasma volume may be reduced in all types of shock, and that the measurement of central venous pressure is useful in assessing the adequacy of the circulating blood volume. Therefore, blood or plasma volume expanders should be used when the principal reason for hypotension or shock is decreased circulating volume.
Because of its vasoconstrictor effect, metaraminol bitartrate injection should be given with caution in heart or thyroid disease, hypertension, or diabetes. Sympathomimetic amines may provoke a relapse in patients with a history of malaria.
DRUG INTERACTIONS
Metaraminol bitartrate injection should be used with caution in digitalized patients, since the combination of digitalis and sympathomimetic amines may cause ectopic arrhythmias.
Monoamine oxidase inhibitors or tricyclic antidepressants may potentiate the action of sympathomimetic amines. Therefore, when initiating pressor therapy in patients receiving these drugs, the initial dose should be small and given with caution. (See WARNINGS.)
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
Studies in animals have not been performed to evaluate the mutagenic or carcinogenic potential of metaraminol bitartrate injection or its potential to affect fertility.
PREGNANCY
Pregnancy Category C.Animal reproduction studies have not been conducted with metaraminol bitartrate injection. It is not known whether metaraminol bitartrate injection can cause fetal harm when given to a pregnant woman or can affect reproduction capacity. Metaraminol bitartrate injection should be given to a pregnant woman only if clearly needed.
Adverse Reactions/Side Effects
Sympathomimetic amines, including metaraminol bitartrate injection, may cause sinus or ventricular tachycardia, or other arrhythmias, especially in patients with myocardial infarction. (See PRECAUTIONS.)
In patients with a history of malaria, these compounds may provoke a relapse.
Abscess formation, tissue necrosis, or sloughing rarely may follow the use of metaraminol bitartrate injection. In choosing the site of injection, it is important to avoid those areas recognized as notsuitable for use of any pressor agent and to discontinue the infusion immediately if infiltration or thrombosis occurs. Although the physician may be forced by the urgent nature of the patient's condition to choose injection sites that are not recognized as suitable, he should, when possible, use the preferred areas of injection. The larger veins of the antecubital fossa or the thigh are preferred to veins in the dorsum of the hand or ankle veins, particularly in patients with peripheral vascular disease, diabetes mellitus, Buerger's disease, or conditions with coexistent hypercoagulability.
Overdosage
Overdosage may result in severe hypertension accompanied by headache, constricting sensation in the chest, nausea, vomiting, euphoria, diaphoresis, pulmonary edema, tachycardia, bradycardia, sinus arrhythmia, atrial or ventricular arrhythmias, cerebral hemorrhage, myocardial infarction, cardiac arrest or convulsions.
Should an excessive elevation of blood pressure occur, it may be immediately relieved by a sympatholytic agent, e.g., phentolamine. An appropriate antiarrhythmic agent may also be required.
The oral LD 50in the rat and mouse is 240 mg/kg and 99 mg/kg, respectively.
Metaraminol Bitartrate Injection Dosage and Administration
Metaraminol bitartrate injection may be given intramuscularly, subcutaneously, or intravenously, the route depending on the nature and severity of the indication.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to use, whenever solution and container permit.
Allow at least 10 minutes to elapse before increasing the dose because the maximum effect is not immediately apparent. When the vasopressor is discontinued, observe the patient carefully as the effect of the drug tapers off, so that therapy can be reinitiated promptly if the blood pressure falls too rapidly. The response to vasopressors may be poor in patients with coexistent shock and acidosis. When indicated, established methods of shock management should be used, such as blood or fluid replacement.
Intramuscular or Subcutaneous Injection(for prevention of hypotension-see INDICATIONS): The recommended dose is 2 to 10 mg (0.2 to 1 mL). As with other agents given subcutaneously, only the preferred sites of injection, as set forth in standard texts, should be used.
Intravenous Infusion(for adjunctive treatment of hypotension - see INDICATIONS): The recommended dose is 15 to 100 mg (1.5 to 10 mL) in 500 mL of Sodium Chloride Injection or 5% Dextrose Injection, adjusting the rate of infusion to maintain the blood pressure at the desired level. Higher concentrations of metaraminol bitartrate injection, 150 to 500 mg per 500 mL of infusion fluid, have been used.
If the patient needs more saline or dextrose solution at a rate of flow that would provide an excessive dose of the vasopressor, the recommended volume of infusion fluid (500 mL) should be increased accordingly. Metaraminol bitartrate injection may also be added to lessthan 500 mL of infusion fluid if a smaller volume is desired.
Compatibility Information
In addition to Sodium Chloride Injection and Dextrose Injection 5%, the following infusion solutions were found physically and chemically compatible with Metaraminol bitartrate injection when 5 mL of Metaraminol bitartrate injection USP 1%, containing metaraminol bitartrate equivalent to 10 mg of metaraminol per mL, was added to 500 mL of infusion solution: Ringer's Injection, Lactated Ringer's Injection, Dextran 6% in Saline**, Normosol
®-R pH 7.4**, and Normosol®-M in D5-W**.
When metaraminol bitartrate injection is mixed with an infusion solution, sterile precautions should be observed. Since infusion solutions generally do not contain preservatives, mixtures should be used within 24 hours.
Direct Intravenous Injection:In severe shock, when time is of great importance, this agent should be given by direct intravenous injection. The suggested dose is 0.5 to 5 mg (0.05 to 0.5 mL), followed by an infusion of 15 to 100 mg (1.5 to 10 mL) in 500 mL of infusion fluid as described previously.
Vials may be sterilized by autoclaving or by immersion in a sterilizing solution.
How is Metaraminol Bitartrate Injection supplied
Metaraminol bitartrate injection USP 1%, containing metaraminol bitartrate equivalent to 10 mg of metaraminol per mL, is a clear, colorless solution and is supplied as follows:
|
NDC number |
Strength |
Vial size |
|
24338-090-01 |
10 mg/mL |
1 X 10 mL Multiple Dose Vial |
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). [See USP Controlled Room Temperature]. Store container in carton until contents have been used. Protect from light. Protect from freezing.
**Product of Abbott Laboratories
Manufactured for:
Azurity Pharmaceuticals, Inc.
Woburn, MA 01801 USA
Manufactured by:
Corden Pharma S.p.A
Caponago, Italy.
March 2024
| METARAMINOL BITARTRATE
metaraminol bitartrate injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Azurity Pharmaceuticals, Inc. (117505635) |
| Registrant - Azurity Pharmaceuticals, Inc. (117505635) |