Dextrose in Ringer's: Package Insert / Prescribing Info
Package insert / product label
Generic name: dextrose, sodium chloride, calcium chloride, and potassium chloride
Dosage form: injection
Drug class: Intravenous nutritional products
Medically reviewed by Drugs.com. Last updated on Mar 25, 2025.
On This Page
Dextrose in Ringer's Description
Each 100 mL of 5% Dextrose in Ringer's Injection contains: Hydrous Dextrose USP 5 g; Sodium Chloride USP 0.86 g; Calcium Chloride Dihydrate USP 0.033 g; Potassium Chloride USP 0.03 g; Water for Injection USP qs
pH: 4.3 (3.5–6.5); Calories per liter: 170
Calculated Osmolarity: 560 mOsmol/liter, hypertonic
Concentration of Electrolytes (mEq/liter): Sodium 147 Potassium 4
Calcium 4.5 Chloride 156
5% Dextrose in Ringer's Injection is sterile, nonpyrogenic, and contains no bacteriostatic or antimicrobial agents.
The formulas of the active ingredients are:
| Ingredients | Molecular Formula | Molecular Weight |
|---|---|---|
| Sodium Chloride USP | NaCl | 58.44 |
| Potassium Chloride USP | KCl | 74.55 |
| Calcium Chloride Dihydrate USP | CaCl2•2H2O | 147.02 |
| Hydrous Dextrose USP | 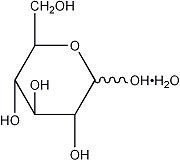 | 198.17 |
The EXCEL® Container is Latex-free; PVC-free; and DEHP-free.
The plastic container is made from a multilayered film specifically developed for parenteral drugs. It contains no plasticizers and exhibits virtually no leachables. The solution contact layer is a rubberized copolymer of ethylene and propylene. The container is nontoxic and biologically inert. The container-solution unit is a closed system and is not dependent upon entry of external air during administration. The container is overwrapped to provide protection from the physical environment and to provide an additional moisture barrier when necessary.
Addition of medication should be accomplished using complete aseptic technique.
The closure system has two ports; the one for the administration set has a tamper evident plastic protector and the other is a medication addition site. Refer to the Directions for Use of the container.
Dextrose in Ringer's - Clinical Pharmacology
5% Dextrose in Ringer's Injection provides electrolytes and calories and is a source of water for hydration. It is capable of inducing diuresis depending on the clinical condition of the patient.
Sodium, the major cation of the extracellular fluid, functions primarily in the control of water distribution, fluid balance, and osmotic pressure of body fluids. Sodium is also associated with chloride and bicarbonate in the regulation of the acid-base equilibrium of body fluid. Potassium, the principal cation of intracellular fluid, participates in carbohydrate utilization and protein synthesis, and is critical in the regulation of nerve conduction and muscle contraction, particularly in the heart.
Chloride, the major extracellular anion, closely follows the metabolism of sodium, and changes in the acid-base balance of the body are reflected by changes in the chloride concentration. Calcium, an important cation, provides the framework of bones and teeth in the form of calcium phosphate and calcium carbonate. In the ionized form, calcium is essential for the functional mechanism of the clotting of blood, normal cardiac function, and regulation of neuromuscular irritability.
Dextrose provides a source of calories. Dextrose is readily metabolized, may decrease losses of body protein and nitrogen, promotes glycogen deposition and decreases or prevents ketosis if sufficient doses are provided.
Indications and Usage for Dextrose in Ringer's
This solution is indicated for use as a source of electrolytes, calories, and water for hydration in adult and pediatric patients.
Contraindications
Solutions containing dextrose may be contraindicated in patients with known allergy to corn products.
This solution is contraindicated where the administration of sodium, potassium, calcium, or chloride could be clinically detrimental.
Warnings
The administration of intravenous solutions can cause fluid and/or solute overload resulting in dilution of serum electrolyte concentrations, overhydration, congested states or pulmonary edema. The risk of dilutional states is inversely proportional to the electrolyte concentration. The risk of solute overload causing congested states with peripheral and pulmonary edema is directly proportional to the electrolyte concentration.
Solutions containing sodium ions should be used with great care, if at all, in patients with congestive heart failure, severe renal insufficiency, and in clinical states in which there is sodium retention with edema.
Solutions containing potassium ions should be used with great care, if at all, in patients with hyperkalemia, severe renal failure, and in conditions in which potassium retention is present.
In patients with diminished renal function, administration of solutions containing sodium or potassium ions may result in sodium or potassium retention.
Solutions containing calcium ions should not be administered simultaneously through the same administration set as blood because of the likelihood of coagulation.
Precautions
General
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations, and acid-base balance during prolonged parenteral therapy or whenever the condition of the patient warrants such evaluation. Significant deviations from normal concentrations may require the use of additional electrolyte supplements, or the use of electrolyte-free dextrose solutions to which individualized electrolyte supplements may be added.
Extraordinary electrolytes losses such as may occur during protracted nasogastric suction, vomiting, diarrhea or gastrointestinal fistula drainage may necessitate additional electrolyte supplementation.
Sodium-containing solutions should be administered with caution to patients receiving corticosteroids or corticotropin, or to other salt-retaining patients.
Potassium therapy should be guided primarily by serial electrocardiograms, especially in patients receiving digitalis. Serum potassium levels are not necessarily indicative of tissue potassium levels.
Care should be exercised in administering solutions containing sodium or potassium to patients with renal or cardiovascular insufficiency, with or without congestive heart failure, particularly if they are postoperative or elderly.
Solutions containing potassium or calcium should be used with caution in the presence of cardiac disease, particularly in the presence of renal disease.
Parenteral calcium should be administered with extreme caution to patients receiving digitalis preparations.
Solutions containing dextrose should be used with caution in patients with overt or known subclinical diabetes mellitus, or carbohydrate intolerance for any reason.
To minimize the risk of possible incompatibilities arising from mixing this solution with other additives that may be prescribed, the final infusate should be inspected for cloudiness or precipitation immediately after mixing, prior to administration, and periodically during administration.
Do not use plastic container in series connection.
If administration is controlled by a pumping device, care must be taken to discontinue pumping action before the container runs dry or air embolism may result. If administration is not controlled by a pumping device, refrain from applying excessive pressure (>300mmHg) causing distortion to the container such as wringing or twisting. Such handling could result in breakage of the container.
This solution is intended for intravenous administration using sterile equipment. It is recommended that intravenous administration apparatus be replaced at least once every 24 hours.
Use only if solution is clear and container and seals are intact.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies with 5% Dextrose in Ringer's Injection have not been performed to evaluate the carcinogenic potential, mutagenic potential, or effects on fertility.
Pregnancy
Teratogenic Effects
Pregnancy Category C
Animal reproduction studies have not been conducted with 5% Dextrose in Ringer's Injection. It is also not known whether 5% Dextrose in Ringer's Injection can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. 5% Dextrose in Ringer's Injection should be given to a pregnant woman only if clearly needed.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when 5% Dextrose in Ringer's Injection is administered to a nursing mother.
Pediatric Use
Safety and effectiveness of 5% Dextrose in Ringer's Injection in pediatric patients have not been established by adequate and well controlled trials, however, the use of dextrose and ringer's solutions in the pediatric population is referenced in the medical literature. The warnings, precautions and adverse reactions identified in the label copy should be observed in the pediatric population.
In very low birth weight infants, excessive or rapid administration of dextrose injection may result in increased serum osmolality and possible hemorrhage.
Geriatric Use
Clinical studies of 5% Dextrose in Ringer's Injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Adverse Reactions/Side Effects
Reactions which may occur because of the solution or the technique of administration include febrile response, infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation and hypervolemia.
Too rapid infusion of hypertonic solutions may cause local pain and venous irritation. Rate of administration should be adjusted according to tolerance. Use of the largest peripheral vein and a well-placed small bore needle is recommended.
Symptoms may result from an excess or deficit of one or more of the ions present in the solution; therefore, frequent monitoring of electrolyte levels is essential.
Hypernatremia may be associated with edema and exacerbation of congestive heart failure due to the retention of water, resulting in an expanded extracellular fluid volume.
Reactions reported with the use of potassium-containing solutions include nausea, vomiting, abdominal pain and diarrhea. The signs and symptoms of potassium intoxication include paresthesias of the extremities, areflexia, muscular or respiratory paralysis, mental confusion, weakness, hypotension, cardiac arrhythmias, heart block, electrocardiographic abnormalities and cardiac arrest. Potassium deficits result in disruption of neuromuscular function, and intestinal ileus and dilatation.
If infused in large amounts, chloride ions may cause a loss of bicarbonate ions, resulting in an acidifying effect.
Abnormally high plasma levels of calcium can result in depression, amnesia, headaches, drowsiness, disorientation, syncope, hallucinations, hypotonia of both skeletal and smooth muscles, dysphagia, arrhythmias and coma. Calcium deficits can result in neuromuscular hyperexcitability, including cramps and convulsions.
Dextrose may be administered at a rate of 0.5 g/kg/hour without producing glycosuria. Hyperglycemia and glycosuria may be a function of rate of administration or metabolic insufficiency. If undetected and untreated this can lead to osmotic dehydration, hyperosmolar coma and death. Appropriate therapy may include slowing of the infusion rate and administration of insulin.
The physician should also be alert to the possibility of adverse reactions to drug additives. Prescribing information for drug additives to be administered in this manner should be consulted.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination if deemed necessary.
Overdosage
In the event of a fluid or solute overload during parenteral therapy, reevaluate the patient's condition, and institute appropriate corrective treatment.
In the event of overdosage with potassium-containing solutions, discontinue the infusion immediately and institute corrective therapy to reduce serum potassium levels.
Treatment of hyperkalemia includes the following:
- Dextrose Injection USP, 10% or 25%, containing 10 units of crystalline insulin per 20 grams of dextrose administered intravenously, 300 to 500 mL per hour.
- Absorption and exchange of potassium using sodium or ammonium cycle cation exchange resin, orally and as retention enema.
- Hemodialysis and peritoneal dialysis. The use of potassium-containing foods or medications must be eliminated. However, in cases of digitalization, too rapid a lowering of plasma potassium concentration can cause digitalis toxicity.
Dextrose in Ringer's Dosage and Administration
This solution is for intravenous use only.
Dosage is to be directed by a physician and is dependent upon age, weight, clinical condition of the patient and laboratory determinations. Frequent laboratory determinations and clinical evaluation are essential to monitor changes in blood glucose and electrolyte concentrations, and fluid and electrolyte balance during prolonged parenteral therapy.
In the average adult, daily requirements of sodium and chloride are met by the infusion of one liter of 0.9% sodium chloride (154 mEq each of sodium and chloride).
There is no specific pediatric dose. The dose is dependent on weight, clinical condition, and laboratory results. Follow recommendations of appropriate pediatric reference text.
As reported in the literature, the dosage and constant infusion rate of intravenous dextrose must be selected with caution in pediatric patients, particularly neonates and low weight infants, because of the increased risk of hyperglycemia/hypoglycemia.
Fluid administration should be based on calculated maintenance or replacement fluid requirements for each patient.
Dextrose may be administered to normal individuals at a rate of 0.5 g/kg/hour without producing glycosuria. At the maximum infusion rate of 0.8 g/kg/hour, approximately 95% of the dextrose is retained.
This solution may be admixed with solutions which contain phosphate or which have been supplemented with phosphate. The presence of calcium ions in this solution should be considered when phosphate is present in the additive solution, in order to avoid precipitation.
Some additives may be incompatible. Consult with pharmacist. When introducing additives, use aseptic techniques. Mix thoroughly. Do not store.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
How is Dextrose in Ringer's supplied
5% Dextrose in Ringer's Injection is supplied sterile and nonpyrogenic in EXCEL® Containers packaged 12 per case.
| NDC | Cat. No. | Size |
|---|---|---|
| 5% Dextrose in Ringer's Injection | ||
| 0264-7781-00 | L7810 | 1000 mL |
Directions for Use of EXCEL® Container
Caution: Do not use plastic container in series connection.
To Open
Tear overwrap down at notch and remove solution container. Check for minute leaks by squeezing solution container firmly. If leaks are found, discard solution as sterility may be impaired. If supplemental medication is desired, follow directions below before preparing for administration.
NOTE: Before use, perform the following checks:
- Inspect each container. Read the label. Ensure solution is the one ordered and is within the expiration date.
- Invert container and carefully inspect the solution in good light for cloudiness, haze, or particulate matter. Any container which is suspect should not be used.
- Use only if solution is clear and container and seals are intact.
Preparation for Administration
- Remove plastic protector from sterile set port at bottom of container.
- Attach administration set. Refer to complete directions accompanying set.
To Add Medication
Warning: Some additives may be incompatible.
To Add Medication Before Solution Administration
- Prepare medication site.
- Using syringe with 18–22 gauge needle, puncture medication port and inner diaphragm and inject.
- Squeeze and tap ports while ports are upright and mix solution and medication thoroughly.
To Add Medication During Solution Administration
- Close clamp on the set.
- Prepare medication site.
- Using syringe with 18–22 gauge needle of appropriate length (at least 5/8 inch), puncture resealable medication port and inner diaphragm and inject.
- Remove container from IV pole and/or turn to an upright position.
- Evacuate both ports by tapping and squeezing them while container is in the upright position.
- Mix solution and medication thoroughly.
- Return container to in use position and continue administration.
PRINCIPAL DISPLAY PANEL - 1000 mL Container Label
5% Dextrose in
Ringer's Injection
REF L7810
NDC 0264-7781-00
1000 mL
EXCEL® CONTAINER
Each 100 mL contains:
Hydrous Dextrose USP 5 g
Sodium Chloride USP 0.86 g
Calcium Chloride•2H2O USP 0.033 g
Potassium Chloride USP 0.03 g
Water for Injection USP qs
pH: 4.3 (3.5-6.5)
Calc. Osmolarity: 560 mOsmol/liter, hypertonic
Electrolytes (mEq/liter):
Na+ 147 K+ 4 Ca++ 4.5 Cl– 156
Sterile, nonpyrogenic. Single dose container.
Do not use in series connection.
For intravenous use only. Use only if solution is
clear and container and seals are intact.
WARNINGS: Do Not Administer Simultaneously
With Blood. Some additives may be incompatible.
Consult with pharmacist. When introducing
additives, use aseptic techniques. Mix thoroughly.
Do not store.
Recommended Storage:
Room temperature (25°C). Avoid excessive heat.
Protect from freezing. See Package Insert.

Rx only

EXCEL is a registered trademark of B. Braun Medical Inc.
B. Braun Medical Inc.
Irvine, CA 92614-5895 USA
1-800-227-2862
www.bbraun.com
Made in USA
Y94-003-022
LD-138-2
Do not remove overwrap until ready for use. After
removing the overwrap, check for minute leaks by
squeezing container firmly. If leaks are found,
discard solution as sterility may be impaired.
EXP
LOT

| DEXTROSE IN RINGERS
dextrose, sodium chloride, calcium chloride, and potassium chloride injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - B. Braun Medical Inc. (002397347) |
More about lvp solution
Patient resources
Professional resources
Other brands
Lactated Ringers Injection, Normosol-R, Extraneal, Delflex, ... +3 more
