Xeomin Dosage
Generic name: BOTULINUM TOXIN TYPE A 50[USP'U]
Dosage form: powder, lyophilized, for solution
Drug class: Skeletal muscle relaxants
Medically reviewed by Drugs.com. Last updated on Dec 19, 2024.
Instructions for Safe Use
The potency Units of XEOMIN for injection are specific to the preparation and assay method utilized. Units of biological activity of XEOMIN cannot be compared to or converted into Units of any other botulinum toxin products assessed with any other specific assay method.
Reconstituted XEOMIN is intended for intramuscular or intra-salivary gland injection only.
Do not exceed the recommended maximum cumulative dose in a treatment session for any indication.
Recommended Dose for Chronic Sialorrhea
Chronic Sialorrhea in Adult Patients
The recommended total dose per treatment session is 100 Units. XEOMIN is injected into the parotid and submandibular glands on both sides (i.e., 4 injection sites per treatment session). The recommended total dose per treatment session is 100 Units. The dose is divided with a ratio of 3:2 between the parotid and submandibular glands (Table 1).
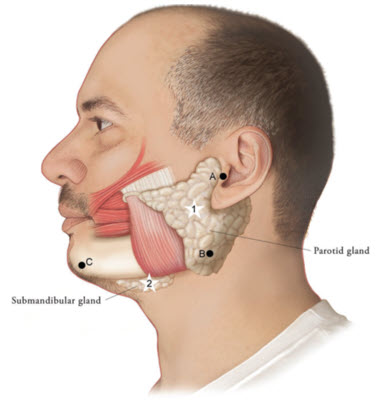
Figure 1: Glands for Injection in Chronic Sialorrhea in Adult Patients
Use the following guidelines if locating salivary glands using anatomic landmarks:
- 1)
- To inject the parotid gland, find the midpoint on the line connecting the tragus and mandible angle (Site A and B, respectively, Figure 1), approximately at the height of the ear lobe. Deliver the injection one finger breadth anterior to this site (Star 1, Figure 1).
- 2)
- To inject the submandibular gland, find the midpoint between the angle of the mandible and the tip of the chin (Site B and C, respectively, Figure 1). Deliver the injection one finger breadth medial to the inferior surface of the mandible at this site (Star 2, Figure 1).
| Gland(s) | Units Per Side | Total |
|---|---|---|
| Parotid gland(s) | 30 Units | 60 Units |
| Submandibular gland(s) | 20 Units | 40 Units |
| Both Glands | 50 Units | 100 Units |
The concentration used in the clinical study after reconstitution was 5 Units/0.1mL. Determine the timing for repeat treatment based on the actual clinical need of the individual patient, and no sooner than every 16 weeks.
Chronic Sialorrhea in Pediatric Patients
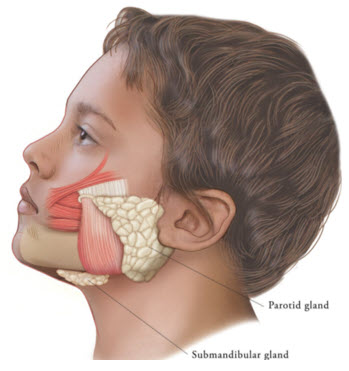
XEOMIN is injected into the parotid and submandibular glands on both sides (i.e., 4 injection sites per treatment session). Ultrasound imaging is recommended to guide needle placement into the salivary glands. The body-weight adjusted dose is divided with a ratio of 3:2 between the parotid and submandibular glands (Table 2). XEOMIN has not been studied in children weighing less than 12 kg.
Figure 2: Glands for Injection in Chronic Sialorrhea in Pediatric Patients
| Body weight | Parotid gland, each side | Submandibular gland, each side | Total dose, both glands, both sides | ||
|---|---|---|---|---|---|
| Dose per gland | Volume per injection | Dose per gland | Volume per injection | ||
| 12 kg or more to less than 15 kg | 6 Units | 0.24 mL | 4 Units | 0.16 mL | 20 Units |
| 15 kg or more to less than 19 kg | 9 Units | 0.36 mL | 6 Units | 0.24 mL | 30 Units |
| 19 kg or more to less than 23 kg | 12 Units | 0.48 mL | 8 Units | 0.32 mL | 40 Units |
| 23 kg or more to less than 27 kg | 15 Units | 0.6 mL | 10 Units | 0.4 mL | 50 Units |
| 27 kg or more to less than 30 kg | 18 Units | 0.72 mL | 12 Units | 0.48 mL | 60 Units |
| 30 kg or more | 22.5 Units | 0.9 mL | 15 Units | 0.6 mL | 75 Units |
The concentration used in the clinical study after reconstitution was 2.5 Units/0.1 mL. Determine the timing for repeat treatment based on the actual clinical need of the individual patient, and no sooner than every 16 weeks.
Recommended Dose for Upper Limb Spasticity
Upper Limb Spasticity in Adult Patients
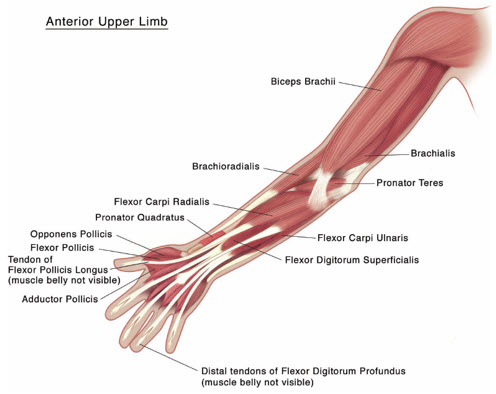
Tailor the dosage, frequency, and number of injection sites to the individual patient based on the size, number, and location of muscles to be treated, severity of spasticity, presence of local muscle weakness, patient's response to previous treatment, and adverse event history with XEOMIN. Administer repeat XEOMIN treatments no sooner than every 12 weeks. In patients not previously treated with a botulinum toxin, begin initial dosing at the low end of the recommended dosing range and titrate as clinically necessary. Most patients in clinical studies were retreated between 12 and 14 weeks.
| Clinical Pattern Muscle |
Units (Range) | Number of injection sites per muscle |
|---|---|---|
| Clenched Fist | ||
| Flexor digitorum superficialis | 25 Units-100 Units | 2 |
| Flexor digitorum profundus | 25 Units-100 Units | 2 |
| Flexed Wrist | ||
| Flexor carpi radialis | 25 Units-100 Units | 1-2 |
| Flexor carpi ulnaris | 20 Units-100 Units | 1-2 |
| Flexed Elbow | ||
| Brachioradialis | 25 Units-100 Units | 1-3 |
| Biceps | 50 Units-200 Units | 1-4 |
| Brachialis | 25 Units-100 Units | 1-2 |
| Pronated Forearm | ||
| Pronator quadratus | 10 Units-50 Units | 1 |
| Pronator teres | 25 Units-75 Units | 1-2 |
| Thumb-in-Palm | ||
| Flexor pollicis longus | 10 Units-50 Units | 1 |
| Adductor pollicis | 5 Units-30 Units | 1 |
| Flexor pollicis brevis/Opponens pollicis | 5 Units-30 Units | 1 |
Figure 3: Muscles Involved In Adult Upper Limb Spasticity

Upper Limb Spasticity in Pediatric Patients, Excluding Spasticity Caused by Cerebral Palsy
Tailor the exact dosage, frequency, and number of injection sites to the individual patient based on size, number and localization of involved muscles; the severity of spasticity; and the presence of local muscle weakness.
The maximum recommended dose is 8 Units/kg, divided among affected muscles, up to a maximum dose of 200 Units per single upper limb. If both upper limbs are treated, do not exceed a total XEOMIN dosage of 16 Units/kg, up to a maximum of 400 Units.
Based on the selected dose, a reconstituted solution at a concentration between 1.25 Units/0.1 mL and 5 Units/0.1 mL is recommended. Determine the timing for repeat treatment based on the clinical need of the patient; administer repeat treatments no sooner than every 12 weeks. Most patients in clinical studies were retreated between 12 and 16 weeks.
Table 4 includes the recommended dose ranges for the treatment of the clinical patterns of flexed elbow, flexed wrist, pronated forearm, clenched fist, and thumb-in-palm.
| Clinical Pattern Muscle |
Dosage | Number of Injection Sites per Muscle | |
|---|---|---|---|
| Range (Units/kg) |
Maximum (Units) |
||
| Flexed Elbow | |||
| Brachioradialis | 1-2 | 50 | 1-2 |
| Biceps | 2-3 | 75 | 1-3 |
| Brachialis | 1-2 | 50 | 1-2 |
| Flexed Wrist | |||
| Flexor carpi radialis | 1 | 25 | 1 |
| Flexor carpi ulnaris | 1 | 25 | 1 |
| Pronated Forearm | |||
| Pronator quadratus | 0.5 | 12.5 | 1 |
| Pronator teres | 1-2 | 50 | 1-2 |
| Clenched Fist | |||
| Flexor digitorum superficialis | 1 | 25 | 1 |
| Flexor digitorum profundus | 1 | 25 | 1 |
| Thumb-in-Palm | |||
| Flexor pollicis longus | 1 | 25 | 1 |
| Adductor pollicis | 0.5 | 12.5 | 1 |
| Flexor pollicis brevis/ opponens pollicis | 0.5 | 12.5 | 1 |
Figure 4: Muscles Injected for Pediatric Upper Limb Spasticity
Recommended Dose for Cervical Dystonia
The recommended initial dose of XEOMIN for cervical dystonia is 120 Units. In a placebo-controlled trial utilizing initial XEOMIN doses of 120 Units and 240 Units, no meaningful difference in effectiveness was demonstrated between the doses. In previously treated patients, their past dose, response to treatment, duration of effect, and adverse event history should be taken into consideration when determining the XEOMIN dose.
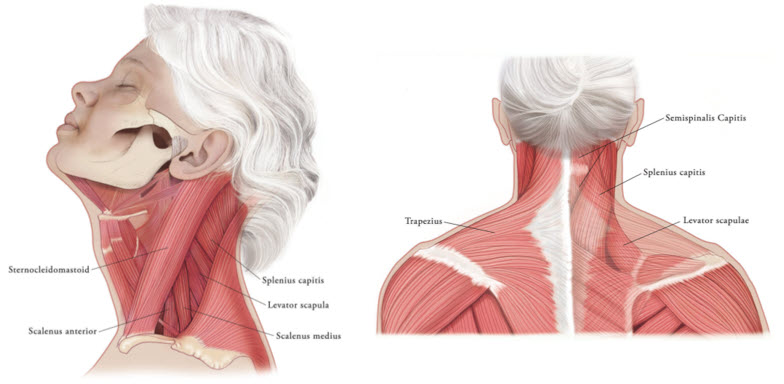
In the treatment of cervical dystonia, XEOMIN is usually injected into the sternocleidomastoid, levator scapulae, splenius capitis, scalenus, and/or the trapezius muscle(s) (see Figure 5). This list is not exhaustive, as any of the muscles responsible for controlling head position may require treatment. The dose and number of injection sites in each treated muscle should be individualized based on the number and location of the muscle(s) to be treated, the degree of spasticity/dystonia, muscle mass, body weight, and response to any previous botulinum toxin injections.
The frequency of XEOMIN repeat treatments should be determined by clinical response, but should generally be no more frequent than every 12 weeks.
Figure 5: Muscles Involved in Cervical Dsytonia
Recommended Dose for Blepharospasm
In treatment-naïve patients, the recommended initial dose of XEOMIN is 50 Units (25 Units per eye). In patients previously treated with a botulinum toxin A, consider their past dose, response to treatment, duration of effect, and adverse event history when determining the XEOMIN dose.
Do not exceed a total XEOMIN dose of 100 Units per treatment session (50 Units per eye).
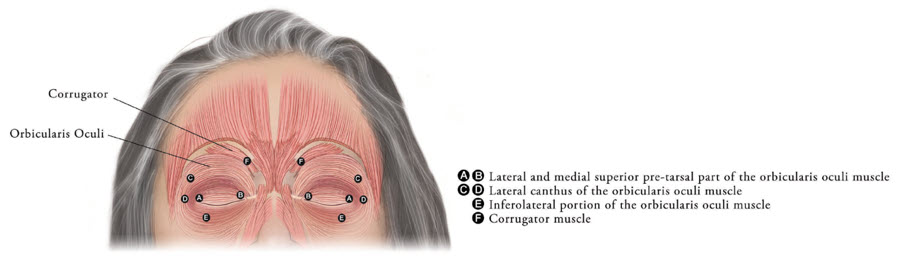
Inject XEOMIN into the lateral and medial orbicularis oculi muscle of the upper lid; lateral canthus and the lateral orbicularis oculi muscle of the lower lid; and the corrugator muscle, if necessary (see Figure 6). The number and location of injections may be changed in response to adverse reactions or based on the patient's response to treatment, but do not exceed a total dose of 50 Units per eye.
Figure 6: Injection Sites for Blepharospasm
Determine the frequency of XEOMIN repeat treatments by clinical response but administer repeat treatments no more frequent than every 12 weeks.
Recommended Dose for Upper Facial Lines (Glabellar Lines, Horizontal Forehead Lines, and Lateral Canthal Lines)
The maximum recommended dose of XEOMIN for simultaneous treatment of upper facial lines [i.e., glabellar lines (GL), horizontal forehead lines (HFL) and lateral canthal lines (LCL)] in adult patients is 64 Units, comprised of 20 Units for GL, 20 Units for HFL, and 24 Units for LCL.
Administer retreatment with XEOMIN no more frequently than every three months.
When not treating upper facial lines (GL, HFL, and LCL) simultaneously in adult patients, refer to the following instructions:
Glabellar Lines
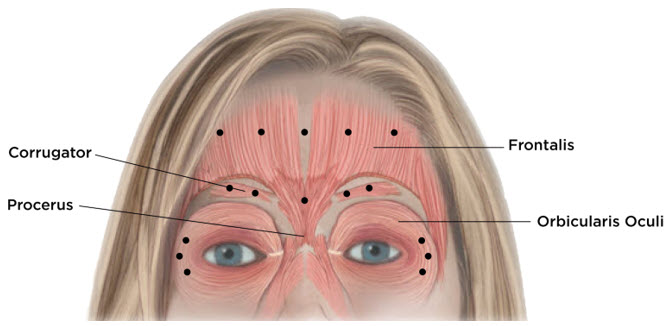
Equally distribute GL treatment to five equal intramuscular injections of 4 Units each. Inject 4 Units of reconstituted XEOMIN intramuscularly into each of 5 sites, 2 in each corrugator muscle and 1 in the procerus muscle for a maximum recommended dose of 20 Units (see Figure 7).
To reduce the complication of ptosis take the following steps:
- Avoid injection near the levator palpebrae superioris, particularly in patients with larger brow depressor complexes.
- Place corrugator injections at least 1 cm above the bony supraorbital ridge.
Horizontal Forehead Lines in Conjunction with Glabellar Lines
Treat HFL in conjunction with GL to minimize the potential for brow ptosis. The maximum recommended dose for treatment of HFL (20 Units) in conjunction with GL (20 Units) is 40 Units.
Equally distribute HFL treatment to 5 horizontally orientated intramuscular injection sites (4 Units each) into the frontalis muscle, at least 2 cm above the orbital rim (see Figure 7).
Lateral Canthal Lines
Inject 4 Units of reconstituted XEOMIN into 3 sites per side (6 total injection sites) in the lateral orbicularis oculi muscle for a total of 12 Units per side (24 Units overall). Place one injection in the horizontal extension of the lateral canthus approximately 1 cm lateral from the bony orbital rim. Place the other two injections approximately 1 cm above and below the area of the first injection (see Figure 7). Give injections with the needle bevel tip up and oriented away from the eye. Avoid injections too close to the zygomaticus major muscle to prevent lip ptosis.
Figure 7: Injection Sites for Upper Facial Lines (Glabellar Lines, Horizontal Forehead Lines, and Lateral Canthal Lines)
Preparation and Reconstitution Technique
Prior to injection, reconstitute each vial of XEOMIN with sterile, preservative-free 0.9% Sodium Chloride Injection, USP. A 20-27 gauge short bevel needle is recommended for reconstitution. Draw up an appropriate amount of preservative-free 0.9% Sodium Chloride Injection, USP into a syringe (see Table 5). Clean the exposed portion of the rubber stopper of the vial with alcohol (70%) prior to insertion of the needle. After vertical insertion of the needle through the rubber stopper, the vacuum will draw the saline into the vial. Gently inject any remaining saline into the vial to avoid foam formation. If the vacuum does not pull the saline into the vial, then XEOMIN must be discarded. Remove the syringe from the vial and mix XEOMIN with the saline by carefully swirling and inverting/flipping the vial – do not shake vigorously. Reconstituted XEOMIN is a clear, colorless solution free of particulate matter. Do not use XEOMIN if the reconstituted solution has a cloudy appearance or contains floccular or particulate matter.
After reconstitution, use XEOMIN for only one injection session and for only one patient. Administer reconstituted XEOMIN within 24 hours after dilution. During this time period, store unused reconstituted XEOMIN in the original container in a refrigerator 2°C -8°C (36°F -46°F) for up to 24 hours until time of use. XEOMIN vials are for single-dose only. Discard any unused portion.
Diluent volumes for reconstitution of XEOMIN are indicated in Table 5.
| Volume of preservative-free 0.9% Sodium Chloride Injection, USP | 50 Unit Vial: Resulting dose in Units per 0.1 mL |
100 Unit Vial: Resulting dose in Units per 0.1 mL |
200 Unit Vial: Resulting dose in Units per 0.1 mL |
|---|---|---|---|
|
|||
| 0.25 mL | 20 Units | - | - |
| 0.5 mL | 10 Units | 20 Units | 40 Units |
| 1 mL | 5 Units | 10 Units | 20 Units |
| 1.25 mL | 4 Units | 8 Units | 16 Units |
| 2 mL | 2.5 Units | 5 Units | 10 Units |
| 2.5 mL | 2 Units | 4 Units | 8 Units |
| 4 mL | 1.25 Units | 2.5 Units | 5 Units |
| 5 mL | 1 Unit | 2 Units | 4 Units |
| 8 mL* | - | 1.25 Units | 2.5 Units |
| 16 mL† | - | - | 1.25 Units |
Administration
Reconstituted XEOMIN is intended for intramuscular or intra-salivary gland injection only.
If proposed injection sites are marked with a pen, DO NOT inject XEOMIN through the pen marks; otherwise a permanent tattooing effect may occur.
For intramuscular injections, the number of injection sites is dependent upon the size of the muscle to be treated and the volume of reconstituted XEOMIN injected.
Inject XEOMIN carefully when injected at sites close to sensitive structures, such as the carotid artery, lung apices, and esophagus. Before administering XEOMIN, the healthcare provider should be familiar with the patient's anatomy and any anatomic alterations, e.g., due to prior surgical procedures.
Chronic Sialorrhea
Chronic Sialorrhea in Adult Patients
Use a sterile needle (e.g., 27-30 gauge (0.30-0.40 mm diameter), 12.5 mm length) for intra-salivary gland administration for the treatment of chronic sialorrhea. Inject XEOMIN close to the center of the gland.
The salivary glands can be located using ultrasound imaging or surface anatomical landmarks.
Chronic Sialorrhea in Pediatric Patients
Use a sterile needle (e.g., 27-30 gauge (0.30-0.40 mm diameter), 12.5 mm length) for intra-salivary gland administration for the treatment of chronic sialorrhea. Inject XEOMIN close to the center of the gland.
Ultrasound guidance is recommended for the localization of the involved salivary glands.
Upper Limb Spasticity
Upper Limb Spasticity in Adult Patients
Use a sterile needle (e.g., 26-gauge (0.45 mm diameter), 37 mm length for superficial muscles; or 22-gauge (0.70 mm diameter), 75 mm length for deeper musculature) in the intramuscular administration in the treatment of upper limb spasticity in adults.
Localization of the involved muscles with electromyographic guidance, nerve stimulation, or ultrasound techniques is recommended.
Upper Limb Spasticity in Pediatric Patients, Excluding Spasticity Caused by Cerebral Palsy
Use a sterile needle (e.g., 30-gauge (0.30 mm diameter), 25 mm length for superficial muscles; or 27-gauge (0.40 mm diameter), 37 mm length for deeper musculature) in the intramuscular administration in the treatment of upper limb spasticity in pediatric patients.
Localization of the involved muscles with techniques such as electromyographic guidance, nerve stimulation, or ultrasound is recommended.
Cervical Dystonia
Use a sterile needle (e.g., 26-gauge (0.45 mm diameter), 37 mm length for superficial muscles; or 22-gauge (0.70 mm diameter), 75 mm length for deeper musculature) in the intramuscular administration in the treatment of cervical dystonia.
Localization of the involved muscles with electromyographic guidance, ultrasound, or nerve stimulation techniques may be useful.
Frequently asked questions
More about Xeomin (incobotulinumtoxinA)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (5)
- Drug images
- Side effects
- During pregnancy
- FDA approval history
- Drug class: skeletal muscle relaxants
- Breastfeeding
- En español
Patient resources
Professional resources
Related treatment guides
See also:
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.