Acetadote: Package Insert / Prescribing Info
Package insert / product label
Generic name: acetylcysteine
Dosage form: injection, solution
Drug class: Antidotes
J Code (medical billing code): J0132 (100 mg, injection)
Medically reviewed by Drugs.com. Last updated on Feb 1, 2023.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
ACETADOTE (acetylcysteine) injection, for intravenous use
Initial U.S. Approval: 2004
Indications and Usage for Acetadote
ACETADOTE is an antidote for acetaminophen overdose indicated to prevent or lessen hepatic injury after ingestion of a potentially hepatotoxic quantity of acetaminophen in patients with an acute ingestion or from repeated supratherapeutic ingestion (RSI) (1).
Acetadote Dosage and Administration
Pre-Treatment Assessment Following Acute Ingestion (2.1):
Obtain a plasma or serum sample to assay for acetaminophen concentration at least 4 hours after ingestion.
- If the time of acetaminophen ingestion is unknown:
- Administer a loading dose of ACETADOTE immediately.
- Obtain an acetaminophen concentration to determine need for continued treatment.
- If the acetaminophen concentration cannot be obtained (or is unavailable or uninterpretable) within the 8-hour time interval after acetaminophen ingestion or there is clinical evidence of acetaminophen toxicity:
- Administer a loading dose of ACETADOTE immediately and continue treatment for a total of three doses over 21 hours.
- If the patient presents more than 8 hours after ingestion and the time of acute acetaminophen ingestion is known:
- Administer a loading dose of ACETADOTE immediately
- Obtain acetaminophen concentration to determine need for continued treatment
- If the patient presents less than 8 hours after ingestion and the time of acute acetaminophen ingestion is known and the acetaminophen concentration is known:
Nomogram for Estimating Potential for Hepatotoxicity from Acute Acetaminophen Ingestion (2.2):
- See Full Prescribing Information for instructions on how to use the nomogram to determine the need for dosing.
Preparation and Storage of Diluted Solution Prior to Administration (2.3):
ACETADOTE is hyperosmolar (2600 mOsmol/L), therefore ACETADOTE
must be diluted in sterile water for injection, 0.45% sodium chloride injection, or 5% dextrose in water injection prior to intravenous administration. See Full Prescribing Information for examples of osmolarity depending on the type of solution and ACETADOTE concentration.
Recommended Adult and Pediatric Dosage (2.4):
- ACETADOTE is for intravenous administration only
- Total dosage of ACETADOTE is 300 mg/kg given intravenously as 3 separate doses and total recommended infusion time for 3 doses is 21 hours
- See Full Prescribing Information for weight-based dosage and weight-based dilution (2.4)
- See Full Prescribing Information for recommendations for continuing ACETADOTE treatment after 21 hours (2.2)
Repeated Supratherapeutic Acetaminophen Ingestion (2.5):
- Obtain acetaminophen concentration and other laboratory tests to guide treatment; Rumack-Matthew nomogram does not apply.
Dosage Forms and Strengths
Injection: 6 grams/30 mL (200 mg/mL) in a single-dose vial (3)
Contraindications
Patients with a previous hypersensitivity reaction to acetylcysteine (4)
Warnings and Precautions
- Hypersensitivity Reactions, Including Hypotension, Wheezing, Shortness of Breath and Bronchospasm: Observe patients during and after the infusion; immediately discontinue infusion if a serious reaction occurs and initiate appropriate treatment. ACETADOTE infusion may be carefully restarted after treatment of hypersensitivity has been initiated (5.1).
- Fluid Overload: Total volume administered should be reduced for patients weighing less than 40 kg and for those requiring fluid restriction (5.2).
Adverse Reactions/Side Effects
Most common adverse reactions (> 2%) are rash, urticaria/facial flushing and pruritus (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Cumberland Pharmaceuticals Inc. at 1-877-484-2700 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 10/2021
Full Prescribing Information
1. Indications and Usage for Acetadote
ACETADOTE is indicated to prevent or lessen hepatic injury after ingestion of a potentially hepatotoxic quantity of acetaminophen in patients with acute ingestion or from repeated supratherapeutic ingestion (RSI).
2. Acetadote Dosage and Administration
2.1 Pre-Treatment Assessment and Testing Following Acute Acetaminophen Ingestion
The following recommendations are related to acute acetaminophen ingestion. For recommendations related to repeated supratherapeutic exposure see Dosage and Administration (2.5).
- Assess the history and timing of acetaminophen ingestion as an overdose.
- The reported history of the quantity of acetaminophen ingested as an overdose is often inaccurate and is not a reliable guide to therapy.
- Obtain the following laboratory tests to monitor hepatic and renal function and electrolyte and fluid balance: aspartate aminotransferase (AST), alanine aminotransferase (ALT), bilirubin, international normalized ratio (INR), creatinine, blood urea nitrogen (BUN), blood glucose, and electrolytes.
- Obtain a plasma or serum sample to assay for acetaminophen concentration at least 4 hours after ingestion. Acetaminophen concentrations obtained earlier than 4 hours post-ingestion may be misleading as they may not represent maximum acetaminophen concentrations.
- If the time of acute acetaminophen ingestion is unknown:
- If the acetaminophen concentration cannot be obtained (or is unavailable or uninterpretable) within the 8-hour time interval after acetaminophen ingestion or there is clinical evidence of acetaminophen toxicity:
- Administer a loading dose of ACETADOTE immediately and continue treatment for a total of three doses over 21 hours [see Dosage and Administration (2.4)].
- If the patient presents more than 8 hours after ingestion and the time of acute acetaminophen ingestion is known:
- If the patient presents less than 8 hours after ingestion and the time of acute acetaminophen ingestion is known and the acetaminophen concentration is known:
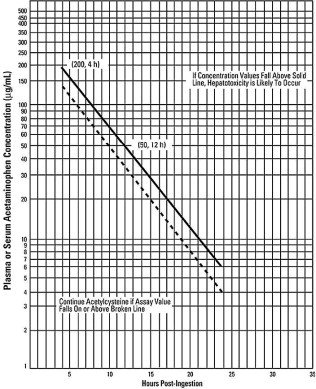
2.2 Nomogram for Estimating Potential for Hepatoxicity from Acute Acetaminophen Ingestion and Need for ACETADOTE Treatment
ACETADOTE is an antidote for acetaminophen overdose. The critical ingestion-treatment interval for maximal protection against severe hepatic injury is between 0 – 8 hours. Efficacy diminishes progressively after 8 hours and treatment initiation between 15 and 24 hours post-ingestion of acetaminophen yields limited efficacy. However, it does not appear to worsen the condition of patients and it should not be withheld, since the reported time of ingestion may not be correct.
If the timing of the acute acetaminophen ingestion is known and the results of the acetaminophen assay are available within 8 hours:
- Refer to the Rumack-Matthew nomogram (see Figure 1) to determine whether or not to initiate treatment with ACETADOTE.
- Initiation of ACETADOTE depends on the plasma or serum acetaminophen concentration and also the clinical presentation of the patient.
The nomogram may underestimate the hepatotoxicity risk in patients with chronic alcoholism, malnutrition, or CYP2E1 enzyme inducing drugs (e.g., isoniazid), and consideration should be given to treating these patients even if the acetaminophen concentrations are in the nontoxic range.
Loading dose
For patients whose acetaminophen concentrations are at or above the “possible” toxicity line (dotted line in nomogram):
- Administer a loading dose of ACETADOTE [see Dosage and Administration (2.4)].
For patients with an acute overdose from an extended-release acetaminophen, if the acetaminophen concentration at 4 hours post ingestion is below the possible toxicity line then obtain a second sample for acetaminophen concentration 8 to 10 hours after the acute ingestion. If the second value is at or above the “possible” toxicity line (dotted line in nomogram):
- Administer a loading dose of ACETADOTE [see Dosage and Administration (2.4)].
For patients whose values are below the “possible” toxicity line, but time of ingestion was unknown or sample was obtained less than 4 hours after ingestion:
- Administer a loading dose of ACETADOTE [see Dosage and Administration (2.4)].
For patients whose values are below the “possible” toxicity line and time of ingestion is known and the sample was obtained more than 4 hours after ingestion, do not administer ACETADOTE because there is minimal risk of hepatotoxicity.
Figure 1. Rumack-Matthew Nomogram for Estimating Potential for Hepatoxicity for Acetaminophen Posioning – Plasma or Serum Acetaminophen Concentration versus Time (hours) Post-acetaminophen Ingestion
(Adapted from Rumack and Matthew, Pediatrics 1975; 55: 871-876)
Maintenance Dose
Determine need for continued treatment with ACETADOTE after the loading dose. Choose ONE of the following based on the acetaminophen concentration:
The acetaminophen concentration is above the possible toxicity line according to the nomogram (see Figure 1):
- Continue ACETADOTE treatment with the maintenance dose for a total of three separate doses over an infusion period of 21 hours [see Dosage and Administration (2.4)].
- Monitor hepatic and renal function and electrolytes throughout treatment.
The acetaminophen concentration could not be obtained:
- Continue ACETADOTE treatment with the maintenance dose for a total of three separate doses over an infusion period of 21 hours [see Dosage and Administration (2.4)].
- Monitor hepatic and renal function and electrolytes throughout treatment.
For patients whose acetaminophen concentration is below the “possible” toxicity line (see Figure 1) and time of ingestion is known and the sample was obtained more than 4 hours after ingestion:
- Discontinue ACETADOTE.
The acetaminophen concentration was in the non-toxic range, but time of ingestion was unknown or less than 4 hours:
- Obtain a second sample for acetaminophen concentration and consider the patient's clinical status to decide whether or not to continue ACETADOTE treatment.
- If there is any uncertainty as to patient's risk of developing hepatotoxicity, it is recommended to administer a complete treatment course.
Continued Therapy After Completion of Loading and Maintenance Doses
In cases of suspected massive overdose, or with concomitant ingestion of other substances, or in patients with preexisting liver disease; the absorption and/or the half-life of acetaminophen may be prolonged. In such cases, consideration should be given to the need for continued treatment with ACETADOTE beyond a total of three separate doses over a 21-hour infusion period.
Acetaminophen levels and ALT/AST and INR should be checked after the last maintenance dose. If acetaminophen levels are still detectable, or if the ALT/AST are still increasing or the INR remains elevated; dosing should be continued and the treating physician should contact a US regional poison center at 1-800-222-1222, alternatively, a “special health professional assistance line for acetaminophen overdose” at 1-800-525-6115 for assistance with dosing recommendations, or 1-877-484-2700 for additional information.
2.3 Preparation and Storage of ACETADOTE Diluted Solution Prior to Administration
Because ACETADOTE is hyperosmolar (2600 mOsmol/L), ACETADOTE must be diluted in sterile water for injection, 0.45% sodium chloride injection (1/2 normal saline), or 5% dextrose in water prior to intravenous administration [see Warnings and Precautions (5.2)]. Dilution in these three solutions results in different osmolarity of the solution for intravenous administration (see Table 1 for examples of different osmolarity of the solution depending on the type of solution and the ACETADOTE concentration).
Visually inspect for particular matter and discoloration prior to administration. The color of the diluted solution ranges from colorless to a slight pink or purple once the stopper is punctured (the color change does not affect the quality of the product). The diluted solution can be stored for 24 hours at room temperature. Discard unused portion. If a vial was previously opened, do not use for intravenous administration.
|
* Adjust osmolarity to a physiologically safe level (generally not less than 150 mOsmol/L in pediatric patients). |
|||
| ACETADOTE Concentration | Osmolarity | ||
| Sterile Water for Injection | ½ Normal Saline | D5W | |
| 7 mg/mL | 91 mOsmol/L* | 245 mOsmol/L | 343 mOsmol/L |
| 24 mg/mL | 312 mOsmol/L | 466 mOsmol/L | 564 mOsmol/L |
2.4 Recommended Dosage in Adults and Pediatrics for Acute Acetaminophen Ingestion
ACETADOTE is for intravenous administration only.
Dosage Regimen
The total recommended dosage of ACETADOTE is 300 mg/kg given intravenously as 3 separate, sequential doses (i.e., 3-bag method to administer the loading, second, and third doses). The total recommended infusion time for 3 doses is 21 hours. For the recommended weight-based dosage and weight-based dilution in patients who weigh:
|
* Dilute ACETADOTE in one of the following three solutions: sterile water for injection, 0.45% sodium chloride injection, or 5% dextrose in water. |
||||||
|
** Recommended dosing for those less than 5 kg has not been studied. |
||||||
| Body Weight | Bag 1 (Loading Dose)
150 mg/kg in 3 mL/kg of diluent* infused over 1 hour | Bag 2 (Second Dose)
50 mg/kg in 7 mL/kg of diluent* infused over 4 hours | Bag 3 (Third Dose)
100 mg/kg diluted in 14 mL/kg of diluent* infused over 16 hours |
|||
| Loading Dose | Diluent Volume | Second Dose | Diluent Volume | Third Dose | Diluent Volume |
|
| 5 kg** | 750 mg | 15 mL | 250 mg | 35 mL | 500 mg | 70 mL |
| 10 kg | 1,500 mg | 30 mL | 500 mg | 70 mL | 1,000 mg | 140mL |
| 15 kg | 2,250 mg | 45 mL | 750 mg | 105 mL | 1,500 mg | 210 mL |
| 20 kg | 3,000 mg | 60 mL | 1,000mg | 140 mL | 2,000 mg | 280 mL |
|
* Dilute ACETADOTE in one of the following three solutions: sterile water for injection, 0.45% sodium chloride injection, or 5% dextrose in water. |
|||
| Body Weight | Bag 1 (Loading Dose)
150 mg/kg in 100 mL of diluent* infused over 1 hour | Bag 2 (Second Dose)
50 mg/kg in 250 mL of diluent* infused over 4 hours | Bag 3 (Third Dose)
100 mg/kg in 500 mL of diluent* infused over 16 hours |
| 21 kg | 3,150 mg | 1,050 mg | 2,100 mg |
| 30 kg | 4,500 mg | 1,500 mg | 3,000 mg |
| 40 kg | 6,000 mg | 2,000 mg | 4,000 mg |
|
* Dilute ACETADOTE in one of the following three solutions: sterile water for injection, 0.45% sodium chloride injection, or 5% dextrose in water. |
|||
|
** No specific studies have been conducted to evaluate the necessity of dose adjustments in patients weighing over 100 kg. Limited information is available regarding the dosing requirements of patients that weigh more than 100 kg. |
|||
| Body Weight | Bag 1 (Loading Dose)
150 mg/kg in 200 mL of diluent1 infused over 1 hour | Bag 2 (Second Dose)
50 mg/kg in 500 mL of diluent1 infused over 4 hours | Bag 3 (Third Dose)
100 mg/kg in 1000 mL of diluent1 infused over 16 hours |
| 41 kg | 6,150 mg | 2,050 mg | 4,100 mg |
| 50 kg | 7,500 mg | 2,500 mg | 5,000 mg |
| 60 kg | 9,000 mg | 3,000 mg | 6,000 mg |
| 70 kg | 10,500 mg | 3,500 mg | 7,000 mg |
| 80 kg | 12,000 mg | 4,000 mg | 8,000 mg |
| 90 kg | 13,500 mg | 4,500 mg | 9,000 mg |
| ≥ 100 kg** | 15,000 mg | 5,000 mg | 10,000 mg |
2.5 Recommendations for Repeated Supratherapeutic Acetaminophen Ingestion
Repeated supratherapeutic acetaminophen ingestion (RSI) is an ingestion of acetaminophen at dosages higher than those recommended for extended periods of time. The risk of hepatotoxicity and the recommendations for treatment of acute acetaminophen ingestion (i.e., the Rumack-Matthew nomogram) do not apply to patients with RSI. Therefore, obtain the following information to guide ACETADOTE treatment for RSI:
- Acetaminophen serum or plasma concentrations. A reported history of the quantity of acetaminophen ingested is often inaccurate and is not a reliable guide to therapy.
- Laboratory tests to monitor hepatic and renal function and electrolyte and fluid balance: AST, ALT, bilirubin, INR, creatinine, BUN, blood glucose, and electrolytes.
For specific ACETADOTE dosage and administration information in patients with RSI, consider contacting your regional poison center at 1-800-222-1222, or alternatively, a special health professional assistance line for acetaminophen overdose at 1-800-525-6115.
3. Dosage Forms and Strengths
Injection: 200 mg/mL (6 grams of acetylcysteine in 30 mL) in a single-dose vial.
4. Contraindications
ACETADOTE is contraindicated in patients with a previous hypersensitivity reaction to acetylcysteine [see Warnings and Precautions (5.1)].
5. Warnings and Precautions
5.1 Hypersensitivity Reactions
Serious acute hypersensitivity reactions during acetylcysteine administration including rash, hypotension, wheezing, and/or shortness of breath, have been observed in patients receiving intravenous acetylcysteine for acetaminophen overdose and occurred soon after initiation of the infusion [see Adverse Reactions (6.1)]. If a severe hypersensitivity reaction occurs, immediately stop the infusion of ACETADOTE and initiate appropriate treatment.
One patient with asthma developed bronchospasm and died after intravenous administration of acetylcysteine. ACETADOTE should be used with caution in patients with asthma, or where there is a history of bronchospasm. Patients with asthma should be closely monitored during initiation of ACETADOTE therapy and throughout ACETADOTE therapy.
Acute flushing and erythema of the skin may occur in patients receiving acetylcysteine intravenously. These reactions usually occur 30 to 60 minutes after initiating the infusion and often resolve spontaneously despite continued infusion of acetylcysteine. If a reaction to acetylcysteine involves more than simply flushing and erythema of the skin, it should be treated as a hypersensitivity reaction.
Management of less severe hypersensitivity reactions should be based upon the severity of the reaction and include temporary interruption of the infusion and/or administration of antihistaminic drugs. The ACETADOTE infusion may be carefully restarted after treatment of the hypersensitivity symptoms has been initiated; however, if the hypersensitivity reaction returns upon re-initiation of treatment or increases in severity, ACETADOTE should be discontinued and alternative patient management should be considered.
5.2 Fluid Overload
The total volume of ACETADOTE administered should be adjusted for patients less than 40 kg and for those requiring fluid restriction. To avoid fluid overload, the volume of diluent should be reduced as needed [see Dosage and Administration (2)]. If volume is not adjusted fluid overload can occur, potentially resulting in hyponatremia, seizure and death.
Intravenous administration of ACETADOTE can cause fluid overload, potentially resulting in hyponatremia, seizure and death. To avoid fluid overload, use the recommended dilution shown in Tables 2, 3 and 4 [see Dosage and Administration (2.4)].
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In the literature the most frequently reported adverse reactions attributed to intravenous acetylcysteine administration were rash, urticaria and pruritus. The frequency of adverse reactions has been reported to be between 0.2% and 21%, and they most commonly occur during the initial loading dose of acetylcysteine.
Loading Dose/Infusion Rate Study
In a randomized, open-label, multi-center clinical study conducted in Australia in patients with acetaminophen poisoning, the rates of hypersensitivity reactions between a 15-minute and 60-minute intravenous infusion for the 150 mg/kg loading dose of acetylcysteine were compared.
The incidence of drug-related adverse reactions occurring within the first 2 hours following acetylcysteine administration is presented in Table 5. Overall, 17% of patients developed an acute hypersensitivity reaction (18% in the 15-minute infusion group; 14% in the 60-minute infusion group) [see Warnings and Precautions (5.1), Clinical Studies (14)].
|
Unkn= Unknown; NOS= not otherwise specified |
||||||||
| Treatment Group | 15-minutes | 60-minutes | ||||||
| Number of Patients | n=109 | n=71 | ||||||
| Cardiac disorders | 5 (5%) | 2 (3%) | ||||||
| Severity: | Unkn | Mild | Moderate | Severe | Unkn | Mild | Moderate | Severe |
| Tachycardia NOS | 4 (4%) | 1 (1%) | 2 (3%) | |||||
| Gastrointestinal disorders | 16 (15%) | 7 (10%) | ||||||
| Severity: | Unkn | Mild | Moderate | Severe | Unkn | Mild | Moderate | Severe |
| Nausea | 1 (1%) | 6 (6%) | 1 (1%) | 1 (1%) | ||||
| Vomiting NOS | 2 (2%) | 11 (10%) | 2 (3%) | 4 (6%) | ||||
| Immune System Disorders | 20 (18%) | 10 (14%) | ||||||
| Severity: | Unkn | Mild | Moderate | Severe | Unkn | Mild | Moderate | Severe |
| Hypersensitivity reaction | 2 (2%) | 6 (6%) | 11 (10%) | 1 (1%) | 4 (6%) | 5 (7%) | 1 (1%) | |
| Respiratory, thoracic and mediastinal disorders | 2 (2%) | 2 (3%) | ||||||
| Severity: | Unkn | Mild | Moderate | Severe | Unkn | Mild | Moderate | Severe |
| Pharyngitis | 1 (1%) | |||||||
| Rhinorrhea | 1 (1%) | |||||||
| Rhonchi | 1 (1%) | |||||||
| Throat tightness | 1 (1%) | |||||||
| Skin & subcutaneous tissue disorders | 6 (6%) | 5 (7%) | ||||||
| Severity: | Unkn | Mild | Moderate | Severe | Unkn | Mild | Moderate | Severe |
| Pruritus | 1 (1%) | 2 (3%) | ||||||
| Rash NOS | 3 (3%) | 2 (2%) | 3 (4%) | |||||
| Vascular disorders | 2 (2%) | 3 (4%) | ||||||
| Severity: | Unkn | Mild | Moderate | Severe | Unkn | Mild | Moderate | Severe |
| Flushing | 1 (1%) | 1 (1%) | 2 (3%) | 1 (1%) | ||||
Safety Study
A large multi-center study was performed in Canada where data were collected from patients who were treated with intravenous acetylcysteine for acetaminophen overdose between 1980 and 2005. This study evaluated 4709 adult cases and 1905 pediatric cases. The incidence of hypersensitivity reactions in adult (overall incidence 8%) and pediatric (overall incidence 10%) patients is presented in Tables 6 and 7.
| Reaction | Incidence (%) n=4709 |
| Urticaria/Facial Flushing | 6.1% |
| Pruritus | 4.3% |
| Respiratory Symptoms* | 1.9% |
| Edema | 1.6% |
| Hypotension | 0.1% |
| Anaphylaxis | 0.1% |
|
*Respiratory symptoms are defined as presence of any of the following: cough, wheezing, stridor, shortness of breath, chest tightness, respiratory distress, or bronchospasm. |
|
| Reaction | Incidence (%) n=1905 |
| Urticaria/Facial Flushing | 7.6% |
| Pruritus | 4.1% |
| Respiratory Symptoms* | 2.2% |
| Edema | 1.2% |
| Anaphylaxis | 0.2% |
| Hypotension | 0.1% |
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
Limited published case reports and case series of pregnant women exposed to acetylcysteine during various trimesters are not sufficient to inform any drug associated risk. Delaying treatment of acetaminophen overdose may increase the risk of maternal or fetal morbidity and mortality [see Clinical Considerations]. Reproduction studies in rats and rabbits following oral administration of acetylcysteine during the period of organogenesis at doses similar to the total intravenous dose (based on the body surface area) did not cause any adverse effects to the fetus. The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
Reproduction studies have been performed following administration of acetylcysteine during the period of organogenesis in rats at oral doses up to 2000 mg/kg/day (1.1 times the recommended total human intravenous dose of 300 mg/kg based on body surface area comparison) and in rabbits at oral doses up to 1000 mg/kg/day (1.1 times the recommended total human intravenous dose of 300 mg/kg based on body surface area comparison). No adverse developmental outcomes due to acetylcysteine were observed.
8.2 Lactation
Risk Summary
There are no data on the presence of acetylcysteine in human milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for ACETADOTE and any potential adverse effects on the breastfed child from ACETADOTE or from the underlying maternal condition.
8.4 Pediatric Use
Safety and effectiveness of ACETADOTE in pediatric patients have not been established by adequate and well-controlled studies. Use of ACETADOTE in pediatric patients 5 kg and greater is based on clinical practice [see Dosage and Administration (2.4)].
10. Overdosage
An initial 150 mg/kg dose of acetylcysteine for a patient weighting 106 kg was mistakenly calculated as 160 g (a decimal point error resulting in a 10-fold higher than prescribed dose). An hour after the infusion started, the patient complained of generalized heat sensation and body pain and developed widespread urticaria and hypotension. The second acetylcysteine infusion was withheld and the patient was treated for anaphylaxis. Despite treatment the patient subcomed to the acute inflammatory reaction and died.
Single intravenous doses of acetylcysteine at 1000 mg/kg in mice, 2445 mg/kg in rats, 1500 mg/kg in guinea pigs, 1200 mg/kg in rabbits and 500 mg/kg in dogs were lethal. Symptoms of acute toxicity in the animals were ataxia, hypoactivity, labored respiration, cyanosis, loss of righting reflex and convulsions.
11. Acetadote Description
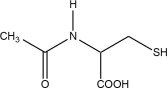
Acetylcysteine injection is an intravenous antidote for the treatment of acetaminophen overdose. Acetylcysteine is the nonproprietary name for the N-acetyl derivative of the naturally occurring amino acid, L-cysteine (N-acetyl-L-cysteine,). The compound is a white crystalline powder, which melts in the range of 104° to 110°C and has a very slight odor.
The molecular formula of the compound is C5H9NO3S, and its molecular weight is 163.2. Acetylcysteine has the following structural formula:
ACETADOTE is supplied as a sterile solution in vials containing 20% w/v (200 mg/mL) acetylcysteine. The pH of the solution ranges from 6.0 to 7.5. ACETADOTE contains the following inactive ingredients: sodium hydroxide (used for pH adjustment), and Water for Injection, USP.
12. Acetadote - Clinical Pharmacology
12.1 Mechanism of Action
Acetylcysteine has been shown to reduce the extent of liver injury following acetaminophen overdose. Acetaminophen doses of 150 mg/kg or greater have been associated with hepatotoxicity. Acetylcysteine probably protects the liver by maintaining or restoring the glutathione levels, or by acting as an alternate substrate for conjugation with, and thus detoxification of, the reactive metabolite of acetaminophen.
12.3 Pharmacokinetics
After a single intravenous dose of acetylcysteine, the plasma concentration of total acetylcysteine declined in a poly-exponential decay manner with a mean terminal half-life (T1/2) of 5.6 hours. The mean clearance (CL) for acetylcysteine was 0.11 liter/hr/kg and renal CL constituted about 30% of the total CL.
Distribution:
The steady-state volume of distribution (Vdss) following administration of an intravenous dose of acetylcysteine was 0.47 liter/kg. The protein binding of acetylcysteine ranges from 66 to 87%.
Elimination
Specific Populations:
Hepatic Impairment:
Following a 600 mg intravenous dose of acetylcysteine to subjects with mild (Child Pugh Class A, n=1), moderate (Child-Pugh Class B, n=4) or severe (Child-Pugh Class C; n=4) hepatic impairment and 6 healthy matched controls, mean T1/2 increased by 80%. Also, the mean CL decreased by 30% and the systemic acetylcysteine exposure (mean AUC) increased 1.6-fold in subjects with hepatic impairment compared to subjects with normal hepatic function. These changes are not considered to be clinically meaningful.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate the carcinogenic potential of acetylcysteine.
Acetylcysteine was not genotoxic in the Ames test or the in vivo mouse micronucleus test. It was, however, positive in the in vitro mouse lymphoma cell (L5178Y/TK+/-) forward mutation test.
Treatment of male rats with acetylcysteine at an oral dose of 250 mg/kg/day for 15 weeks (0.1 times the recommended total human intravenous dose of 300 mg/kg based on body surface comparison) did not affect the fertility or general reproductive performance.
14. Clinical Studies
Loading Dose/Infusion Rate Study
A randomized, open-label, multi-center clinical study was conducted in Australia in patients with acetaminophen poisoning to compare the rates of hypersensitivity reactions between two rates of infusion for the intravenous acetylcysteine loading dose. One hundred nine subjects were randomized to a 15-minute infusion rate and seventy-one subjects were randomized to a 60 minute infusion rate. The loading dose was 150 mg/kg followed by a maintenance dose of 50 mg/kg over 4 hours and then 100 mg/kg over 16 hours. Of the 180 patients, 27% were male and 73% were female. Ages ranged from 15 to 83 years, with the mean age being 30 years (±13.0).
A subgroup of 58 subjects (33 in the 15-minute infusion group; 25 in the 60-minute infusion group) was treated within 8 hours of acetaminophen ingestion. No hepatotoxicity occurred within this subgroup; however, with 95% confidence, the true hepatotoxicity rates could range from 0% to 9% for the 15-minute infusion group and from 0% to 12% for the 60-minute infusion group.
Observational Study
An open-label, observational database contained information on 1749 patients who sought treatment for acetaminophen overdose over a 16-year period. Of the 1749 patients, 65% were female, 34% were male and less than 1% was transgender. Ages ranged from 2 months to 96 years, with 72% of the patients falling in the 16- to 40-year-old age bracket. A total of 399 patients received acetylcysteine treatment. A post-hoc analysis identified 56 patients who (1) were at high or probable risk for hepatotoxicity (APAP greater than 150 mg/L at the four hours line according to the Australian nomogram) and (2) had a liver function test. Of the 53 patients who were treated with intravenous acetylcysteine (300 mg/kg intravenous acetylcysteine administered over 20-21 hours) within 8 hours, two (4%) developed hepatotoxicity (AST or ALT greater than 1000 U/L). Twenty-one of 48 (44%) patients treated with acetylcysteine after 15 hours developed hepatotoxicity. The actual number of hepatotoxicity outcomes may be higher than what is reported here. For patients with multiple admissions for acetaminophen overdose, only the first overdose treated with intravenous acetylcysteine was examined. Hepatotoxicity may have occurred in subsequent admissions.
Evaluable data were available from a total of 148 pediatric patients (less than 16 years of age) who were admitted for poisoning following ingestion of acetaminophen, of whom 23 were treated with intravenous acetylcysteine. There were no deaths of pediatric patients. None of the pediatric patients receiving intravenous acetylcysteine developed hepatotoxicity while two patients not receiving intravenous acetylcysteine developed hepatotoxicity. The number of pediatric patients is too small to provide a statistically significant finding of efficacy; however the results appear to be consistent to those observed for adults.
16. How is Acetadote supplied
ACETADOTE (acetylcysteine) injection is available as a 20% solution (200 mg/mL) in 30 mL single-dose glass vials. Each single dose vial contains 6 g/30mL (200 mg/mL) of ACETADOTE injection. ACETADOTE is sterile and can be used for intravenous administration. It is available as follows:
- 30 mL vials, carton of 4 (NDC 66220-207-30)
Do not use previously opened vials for intravenous administration.
Note: The color of ACETADOTE may turn from essentially colorless to a slight pink or purple once the stopper is punctured. The color change does not affect the quality of the product.
The stopper in the ACETADOTE vial is formulated with a synthetic base-polymer and does not contain Natural Rubber Latex, Dry Natural Rubber, or blends of Natural Rubber.
Store unopened vials at controlled room temperature, 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]
17. Patient Counseling Information
Hypersensitivity Reactions
Advise patients and caregivers that hypersensitivity reactions related to administration and infusion may occur during and after ACETADOTE treatment, including hypotension, wheezing, shortness of breath and bronchospasm [see Warnings and Precautions (5.1)].
For specific treatment information regarding the clinical management of acetaminophen overdose, please contact your regional poison center at 1-800-222-1222, or alternatively, a special health professional assistance line for acetaminophen overdose at 1-800-525-6115.
Manufactured for:
Cumberland Pharmaceuticals Inc.
Nashville, TN 37203
U.S. Patent Nos. 8,148,356, 8,399,445, 8,653,061 8,722,738 and 9,327,028
PRINCIPAL DISPLAY PANEL – Vial Label
30 mL NDC 66220-207-30
Sterile
Acetadote®
(acetylcysteine) Injection
6 g/30 mL
(200 mg/mL)
MUST BE FURTHER DILUTED
PRIOR TO INTRAVENOUS USE
CUMBERLAND®
PHARMACEUTICALS
PRINCIPAL DISPLAY PANEL – Carton Label
4 × 30mL Sterile Vials NDC 66220-207-30
Acetadote®
(acetylcysteine) Injection
6 g/30 mL
(200 mg/mL)
MUST BE FURTHER DILUTED
PRIOR TO INTRAVENOUS USE
CUMBERLAND®
PHARMACEUTICALS
Manufactured for:
Cumberland Pharmaceuticals Inc.
Nashville, TN, USA, 37203
Made in USA
| ACETADOTE
acetylcysteine injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Cumberland Pharmaceuticals Inc. (069532880) |
Frequently asked questions
More about Acetadote (acetylcysteine)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Side effects
- Dosage information
- During pregnancy
- Generic availability
- FDA approval history
- Drug class: antidotes
- Breastfeeding